Latest News
Research & Innovation
Researchers identify Achilles’ heel in lung cancer through common p53 genetic mutation
Mar 9, 2026
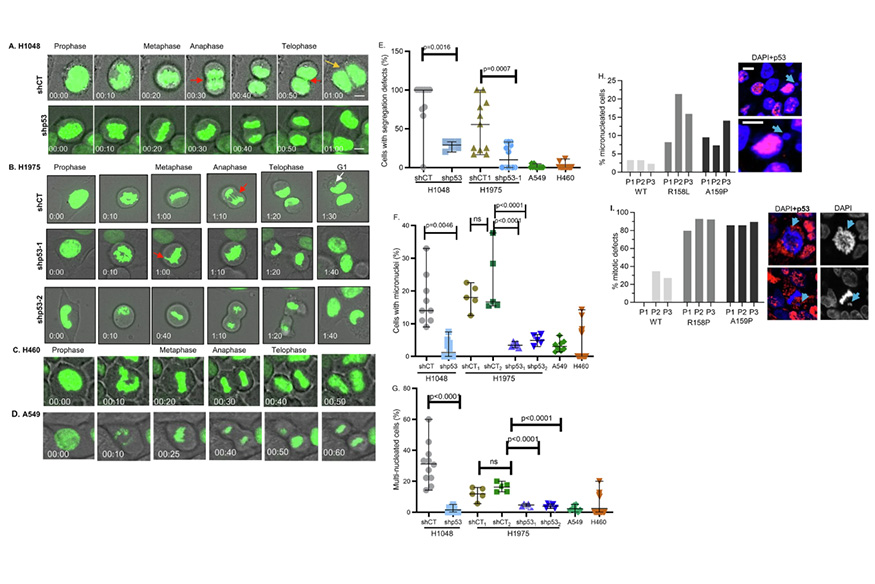 New findings from VCU Massey Comprehensive Cancer Center support an innovative framework for how targeting a new pathway could help to stop the replication of lung tumor cells and thwart tumor growth. This figure includes time-lapse live images and videos from the research. [Singh, S., Gheghiani, L., Velasco, B. et al. Cell Death Differ (2026)]
New findings from VCU Massey Comprehensive Cancer Center support an innovative framework for how targeting a new pathway could help to stop the replication of lung tumor cells and thwart tumor growth. This figure includes time-lapse live images and videos from the research. [Singh, S., Gheghiani, L., Velasco, B. et al. Cell Death Differ (2026)]
A team of researchers at VCU Massey Comprehensive Cancer Center has identified a new pathway through which mutations in the tumor suppressor p53 gene—found very frequently in human tumors—hijack DNA replication in cancer cells. New findings published in Cell Death & Differentiation support an innovative framework for how targeting this new pathway could help to stop the replication of lung tumor cells and thwart tumor growth.
“These findings will be extremely valuable for tumor-specific treatment of a high percentage of cancer patients with a p53 mutation,” said Swati Palit Deb, Ph.D., a member of the Cancer Biology research program at Massey and an associate professor in the Department of Cellular, Molecular and Genetic Medicine at the VCU School of Medicine. “Our study provides compelling evidence that targeting mutant p53 using cell cycle checkpoint inhibitors has the potential to abolish tumors with the p53 mutation, particularly lung tumors.”
Mutations in p53 are very common in all cancers, but they are observed at an even higher rate in lung cancer, appearing in up to 70-90% of tumors for certain forms of disease. A normal p53 gene is a tumor suppressor, meaning its activity stops the formation of tumors. When p53 mutates, it not only loses its tumor suppressor ability, but also gains tumor formation function.
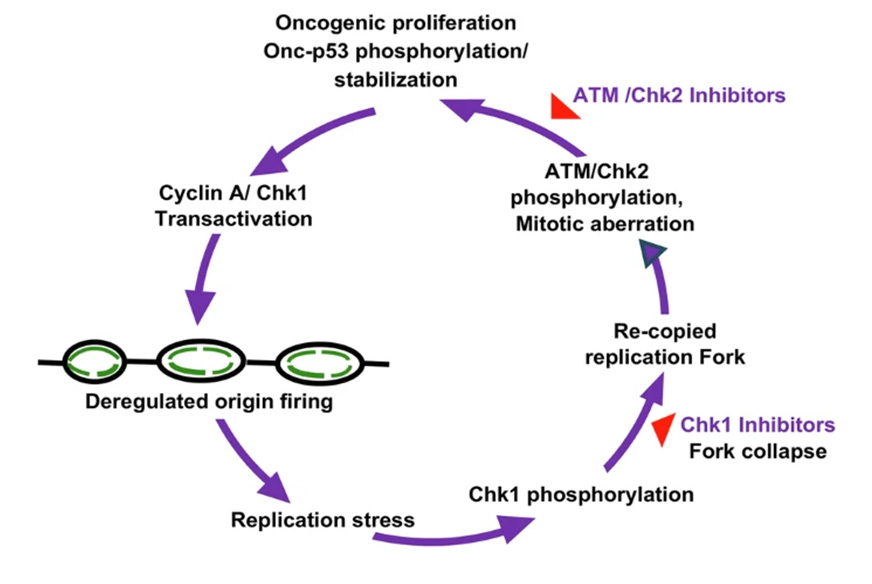 A team of researchers at VCU Massey Comprehensive Cancer Center has identified a new pathway through which mutations in the tumor suppressor p53 gene hijack DNA replication in cancer cells, and establish a feed-forward loop to strengthen its tumor formation ability. [Singh, S., Gheghiani, L., Velasco, B. et al. Cell Death Differ (2026)]
A team of researchers at VCU Massey Comprehensive Cancer Center has identified a new pathway through which mutations in the tumor suppressor p53 gene hijack DNA replication in cancer cells, and establish a feed-forward loop to strengthen its tumor formation ability. [Singh, S., Gheghiani, L., Velasco, B. et al. Cell Death Differ (2026)]
While DNA replication is a key component of cell multiplication and cancer development, Palit Deb and her research team are the first to make discoveries in mutant p53-induced replication stress. Previous research has suggested that mutant p53 binds with other proteins to stabilize itself and accelerate tumor development during replication stress, but Palit Deb’s findings demonstrate that mutant p53 can act alone to stabilize itself, accelerating tumor development.
“During DNA replication, stress usually prevents cell multiplication. The checkpoint pathways tell them they are going too fast. However, mutant p53 is unique in that it activates expression of proteins that allow cells to ignore these pathways and multiply anyway,” said Palit Deb. “We found that when cancer cells with mutant p53 multiply, they make mistakes during cell division but utilize those cellular errors to actually reinforce themselves, initiating a feed-forward loop that drives continued cancer growth.”
Through a previous study published in the Journal of Clinical Investigation, the research team found that checkpoint inhibition collapses DNA replication forks to inhibit tumor growth. The new discovery they uncovered is that tumor-derived p53 mutants stabilize themselves and thus establish a feed-forward loop to strengthen its tumor formation ability. Therefore, combined inhibition of the Chk1 and ATM pathways effectively kills p53-expressing tumor cells, potentially pointing to an effective therapeutic option with lower dosages and reduced toxicity.
View timelapse photos and videos of treating lung cancer cells with ATM and Chk1 inhibitors
To target these replication pathways, Palit Deb has begun screening various drugs using the National Cancer Institute library to determine which one will be the most effective while not interfering with normal cell function.
While this research was conducted on lung cancer models, the implications of their findings could be applied to any cancer with a p53 mutation, leading to cancer cell-specific treatment options for patients facing cancer with p53 mutation.
Palit Deb collaborated on this study with lead author Shilpa Singh, Ph.D., a graduate of the VCU School of Medicine; and additional collaborators with former and current roles at VCU, including Sumitra Deb, Ph.D., Rebecca Frum, Ph.D., Lilia Gheghiani, Ph.D., Steven R. Grossman, M.D., Ph.D., Brandon Velasco, and Brad Windle, Ph.D.
This study was funded by the Commonwealth Health Research Board of Virginia, a pilot project from VCU Massey Comprehensive Cancer Center, multiple grants from the National Cancer Institute, and, in part, by an award from the American Association for Cancer Research.
Written by: Tatiana Del Valle, Blake Belden
Related News
Research & Innovation
Neighborhood factors related to financial stress are linked to worse breast cancer outcomes; interventions could improve survivorshipFeb 18, 2026
Research & Innovation, Clinical Care, Community Engagement
Clinical trials: Shaping the standard of cancer care for the countryFeb 9, 2026
Research & Innovation
How a heart medication could unlock a new targeted approach in lymphomaFeb 4, 2026

Get access to new, innovative care
Treatments in clinical trials may be more effective or have fewer side effects than the treatments that are currently available. With more than 200 studies for multiple types of cancers and cancer prevention, Massey supports a wide array of clinical trials.
Find a provider
Massey supports hundreds of top cancer specialists serving the needs of our patients. Massey’s medical team provides a wealth of expertise in cancer diagnosis, treatment, prevention and symptom management.
